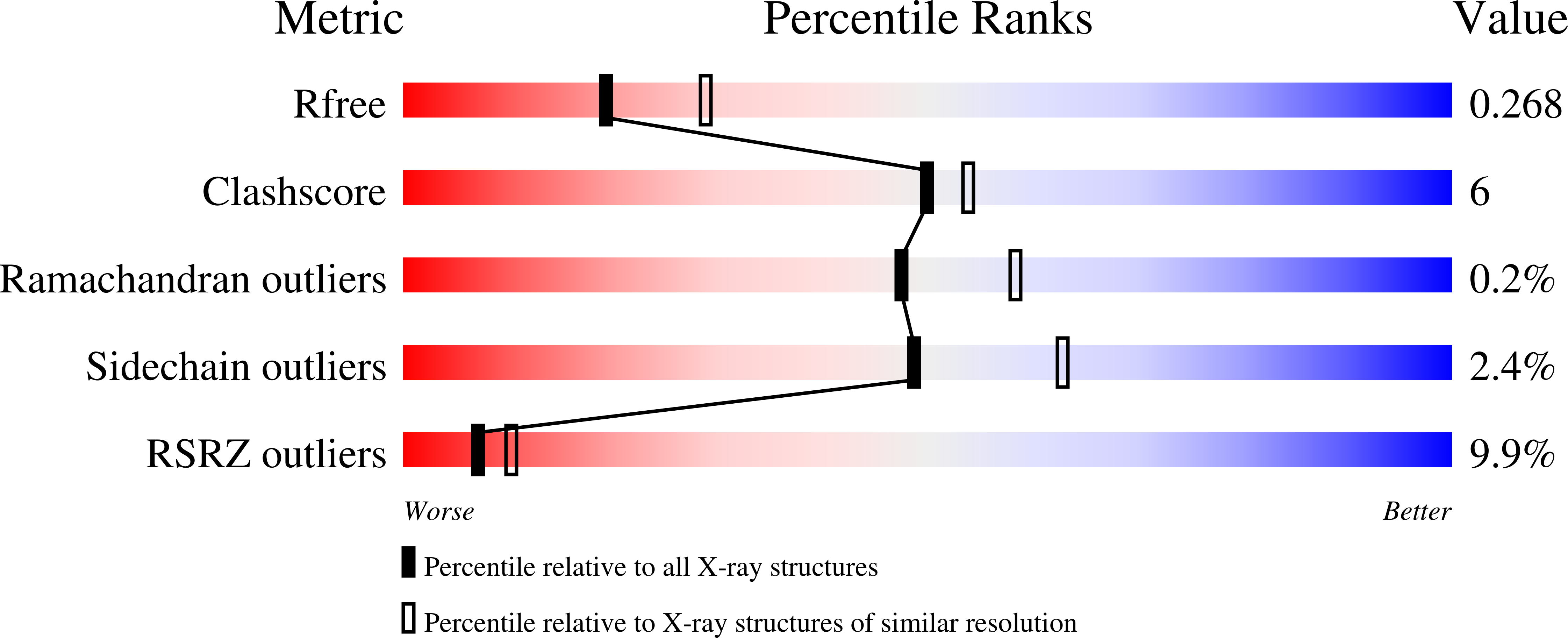
RIG-I contains two N-terminal caspase activation and recruitment domains (CARDs), which are required for interaction with IPS-1 ( Yoneyama et al, 2004), a superfamily 2 helicase/translocase/ATPase (SF2) domain and a C-terminal regulatory/repressor domain (RD Saito et al, 2007).
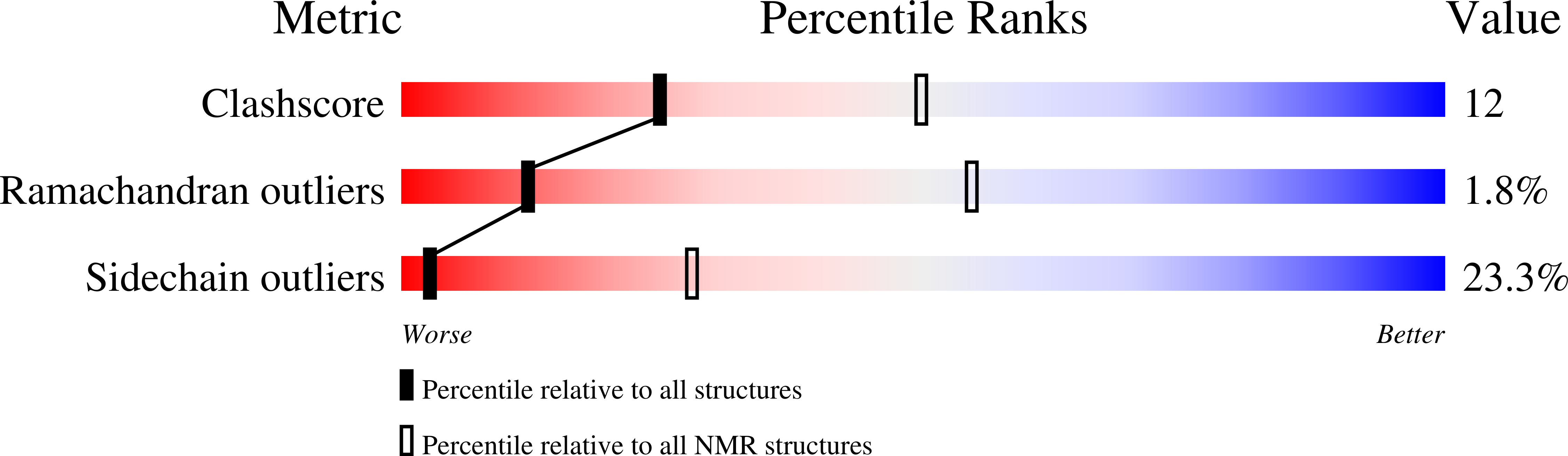
The optimal ligand for RIG-I has been found to be base-paired or double-stranded RNA (dsRNA) molecules containing a 5′ triphosphate (5′-ppp-dsRNA Hornung et al, 2006 Pichlmair et al, 2006 Schlee et al, 2009). RIG-I senses infections from viruses such as hepatitis C virus, Sendai virus, influenza virus, vesicular stomatitis virus, rabies virus and Japanese encephalitis virus ( Kato et al, 2006).Īfter sensing RNA ligands, RIG-I associates with interferon-β promoter stimulator 1 (IPS-1), a process that involves RIG-I ubiquitination and/or interaction with ubiquitin chains ( Gack et al, 2007 Zeng et al, 2010). RIG-I-like receptors (RLRs) sense cytoplasmic viral RNA and comprise RIG-I, MDA5 (melanoma differentiation-associated protein 5) and LGP2 (laboratory of genetics and physiology 2 Kang et al, 2002 Yoneyama et al, 2004 Rothenfusser et al, 2005). In mammals, the formation of PRR–PAMP complexes start signalling cascades that activate the transcription factors nuclear factor-κB and interferon regulatory factors, triggering host defense mechanisms such as the activation of interferon-regulated genes and inflammatory responses. In the innate immune system, pattern recognition receptors (PRRs) distinguish self versus non-self by binding molecular patterns that are present on pathogen-associated molecules (pathogen-associated molecular patterns (PAMPs)) but typically not on host molecules ( Takeuchi & Akira, 2010). The innate immune system is the first line of defence against infections by pathogens. Overall, our results indicate that the activation of RIG-I occurs through an RNA- and ATP-driven structural switch in the SF2 domain. The structure also indicates that the regulatory domain is close to the end of the helical arm, where it is well positioned to recruit 5′-ppp-dsRNA to the SF2 domain. By mutational analysis, we show surface motifs that are critical for dsRNA-stimulated ATPase activity, indicating that dsRNA induces a structural movement that brings domains 1A and 2A/B together to form an active ATPase site. Domains 1A and 2A are maintained in a ‘signal-off’ state with an inactive ATP hydrolysis site by an intriguing helical arm. RIG-I SF2 comprises two RecA-like domains 1A and 2A and a helical insertion domain 2B, which together form a ‘C’-shaped structure. Here we report the crystal structure of superfamily 2 (SF2) ATPase domain of RIG-I in complex with a nucleotide analogue. RIG-I detects cytosolic viral dsRNA with 5′ triphosphates (5′-ppp-dsRNA), thereby initiating an antiviral innate immune response.
